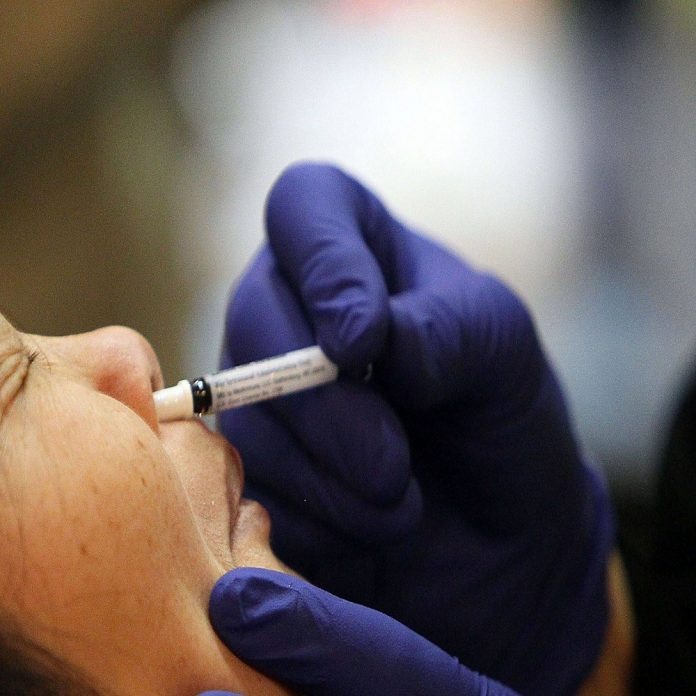
India may soon receive the first Corona nasal vaccine. Nasal vaccination is one that is administered through the nose. Prime Minister Narendra Modi recently stated this in a speech to the nation. The nasal vaccine is expected to arrive in the country by January.
What is Nasal Vaccine and How Does It Work?
We are currently receiving the vaccine through injection into the muscle. Intramuscular vaccination is the name of this vaccine. Nasal vaccines are those that are administered through the nose. It’s called an intranasal vaccine since it’s administered through the nose.
Which Nasal Vaccines Could You Get in India?
BBV154 is the name of the vaccination available in India. This vaccine is being developed in collaboration between Bharat Biotech and Washington University School of Medicine (WUSM). Bharat Biotech has stated that it plans to generate 100 crore vaccine doses by 2022.
What have been the results of the trials thus far?
The initial phase of the vaccine experiment yielded satisfactory results. After receiving the vaccination, none of the volunteers suffered any major side effects. This nasal vaccine was given to 175 participants in four cities, according to the Clinical Trials Registry. Previously, the vaccine was proved to be safe even in pre-clinical studies, meaning it worked well on rats and other laboratory animals. This vaccination produced a considerable number of neutralising antibodies during animal testing.
The results of Phase-2 vaccine studies, according to Bharat Biotech Chairman Krishna Ella, were likewise promising. The vaccine is now being prepared for a Phase 3 experiment. The company applied to the DGCI for Phase-III trials on December 20.
Read More: Get iPhone at very low price in Flipkart Sale, Buy Now Today is the last date!
What distinguishes this vaccination from other vaccines?
In total, eight vaccines have been licensed in the country thus far. All of these vaccines are administered intramuscularly, that is, by injection. BBV154, on the other hand, is an intranasal vaccination. Sputnik, Covishield, and Covaccine are currently being implemented around the country. These three immunizations are given in two doses. BBV154 will only be administered once.
What is the mechanism of action of the nasal vaccine?
Many microorganisms (microscopic viruses) enter the body through the mucosa, including coronaviruses (wet, sticky substances found in the nose, mouth, lungs, and digestive tract). The nasal vaccine activates the immune system directly in the mucosa.
The nasal vaccination, in other words, prepares soldiers to battle where the virus has infiltrated the body. The nasal vaccine causes your body to manufacture immunoglobulin A (igA). IgA is thought to be more effective at preventing infection in the early stages. It protects against both infection and transmission.
What are the advantages of getting a nasal vaccine?
– In India, two doses of the vaccine are being delivered at the moment. After 14 days, the person who has been vaccinated is regarded safe. The nasal vaccine begins to work within 14 days in this case.
– Not only will an effective nasal dose guard against coronavirus, but it will also prevent the disease from spreading. Mild symptoms will be unnoticed by the patient as well. The virus won’t be able to infect other sections of the body, either.
– Because it is a single-dose vaccine, it is simple to track. It also has fewer negative effects than injectable immunisation. Another significant benefit is that needle and syringe waste will be reduced.
Is it going to work on Omicron?
Although more research is needed, preliminary findings suggest that the nasal vaccine is effective against multiple flu virus strains rather than just one.
The nasal vaccine was successful in dealing with respiratory virus in mice, according to a study published in the journal Science Immunology, whereas the intramuscular vaccine had no effect.

